Question 247Steam Plants - Assistant Engineer
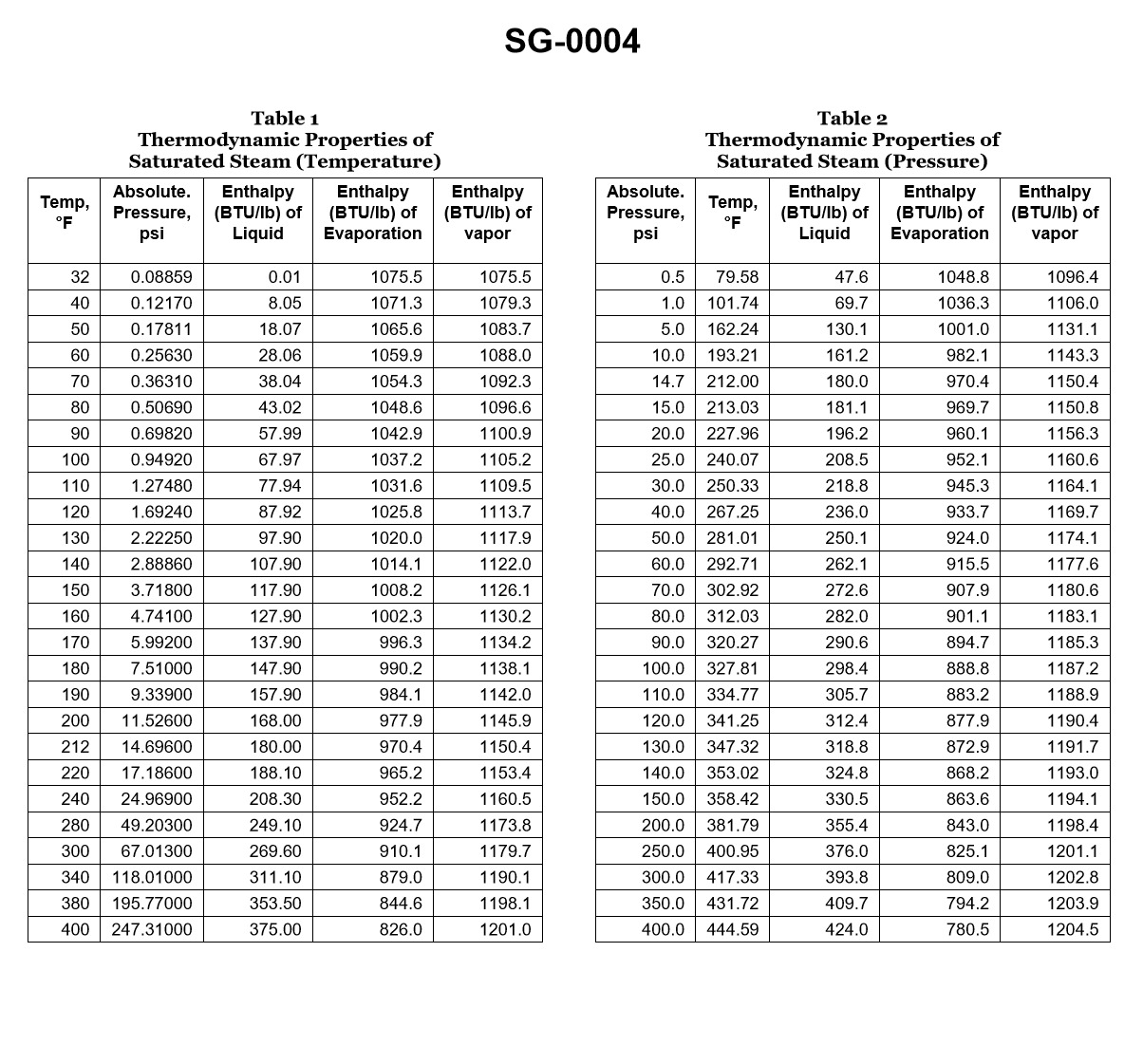
70% to passWith reference to the chart, if a boiler generates saturated steam at 385.3 psig, how much heat per pound was required to change the water into steam if the feed water temperature was initially 104.5°C? Illustration SG-0004
A96.85 BTU
B97.15 BTU
C1016.40 BTU
D1196.45 BTU
AI Explanation
The correct answer is C) 1016.40 BTU. To determine the heat required to change the water into steam, we need to calculate the enthalpy of vaporization (latent heat) at the given pressure of 385.3 psig. At this pressure, the saturated steam temperature is approximately 435°F (223.9°C). Using steam tables or appropriate thermodynamic calculations, the enthalpy of vaporization at this temperature is approximately 1016.40 BTU/lb. This is the amount of heat required to change the water into steam, given the initial feed water temperature of 104.5°C. The other options are incorrect because they do not accurately represent the enthalpy of vaporization at the specified pressure and temperature conditions.
Related Questions
Q477:If there is a sudden drop in the outlet temperature of an uncontrolled superheat... Q528:When a turbine rotor is not rotating during maneuvering, the heat tends to be co... Q49:Scale in the air ejector first-stage nozzle could cause a decrease in the ______... Q418:If a ship is to be laid up for an indefinite period, the steam side of the main ... Q213:The tube sheets installed in a fire-tube auxiliary boiler are normally connected...
Ready to test your knowledge?
Take a Steam Plants - Assistant Engineer Practice ExamOfficial Resources