Question 248Steam Plants - Assistant Engineer
70% to passAccording to the data given in the illustration, which of the following would be the physical state of the fluid at a gage vacuum of 29.00 inches Hg, and 85.21 degrees Fahrenheit? Illustration SG-0026
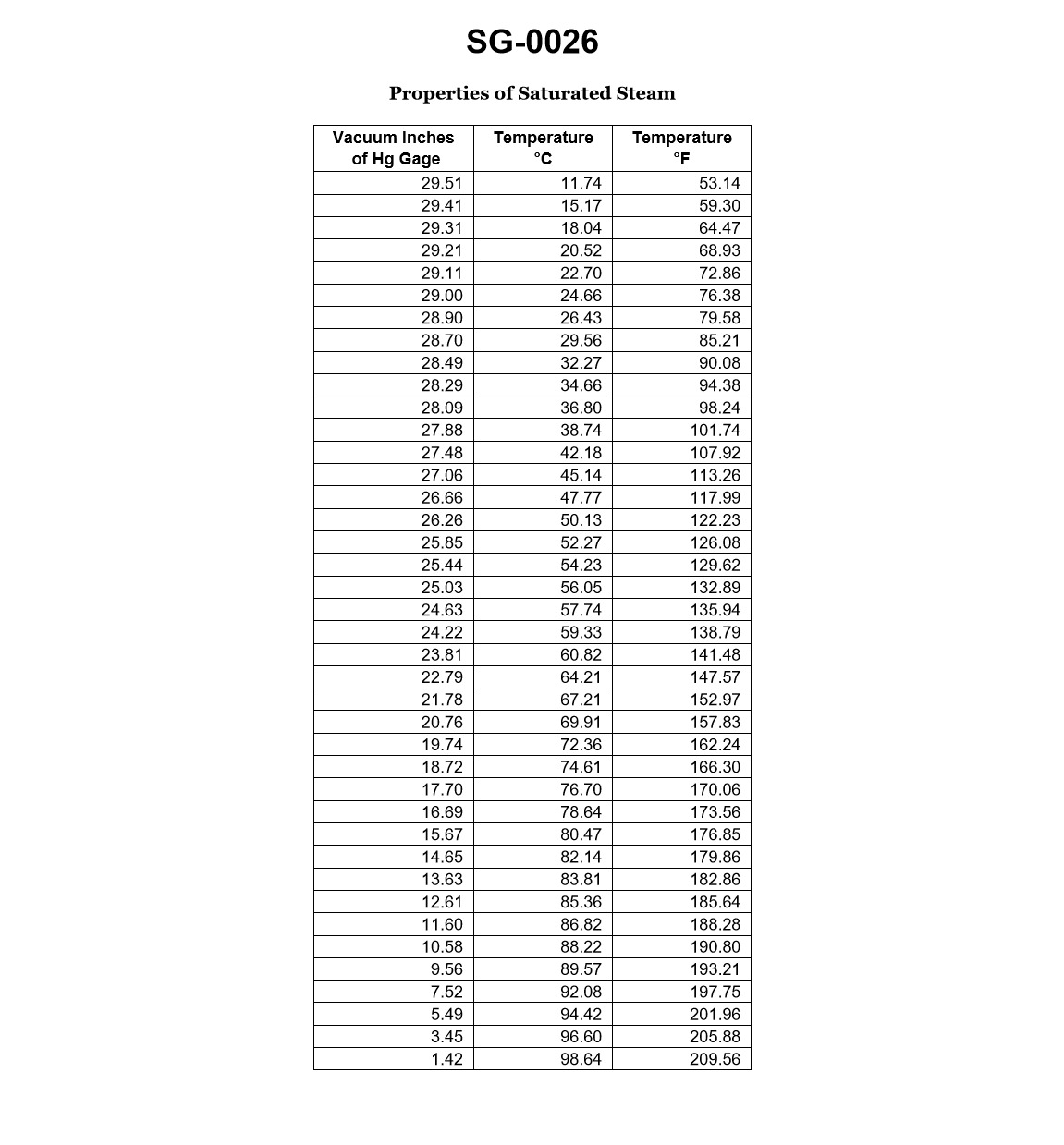
ASub cooled liquid.
BSaturated liquid.
CSuperheated vapor.
DMixture of saturated liquid and vapor.
AI Explanation
The correct answer is C) Superheated vapor. At a gage vacuum of 29.00 inches Hg and a temperature of 85.21 degrees Fahrenheit, the fluid would be in a superheated vapor state. This is because the temperature and pressure conditions are above the saturation point of the fluid, meaning the fluid is in a superheated state and not a saturated liquid or a mixture of liquid and vapor. The other options are incorrect because a gage vacuum of 29.00 inches Hg would not result in a subcooled liquid (A) or a saturated liquid (B), and the temperature and pressure conditions are not within the two-phase region, ruling out a mixture of saturated liquid and vapor (D).
Related Questions
Q170:Air accumulated in the inter condenser of the air ejector assembly is discharged... Q644:Fine metallic particles, which may originate from wear or failure of the lube oi... Q285:The purpose of try-cocks used on an auxiliary boiler is to _______________. Q320:As found in a basic pneumatic automatic combustion control system, the function ... Q456:The viscosity of a residual fuel oil is measured in Saybolt _______________.
Ready to test your knowledge?
Take a Steam Plants - Assistant Engineer Practice ExamOfficial Resources