Question 231Steam Plants - 1st Asst/Chief
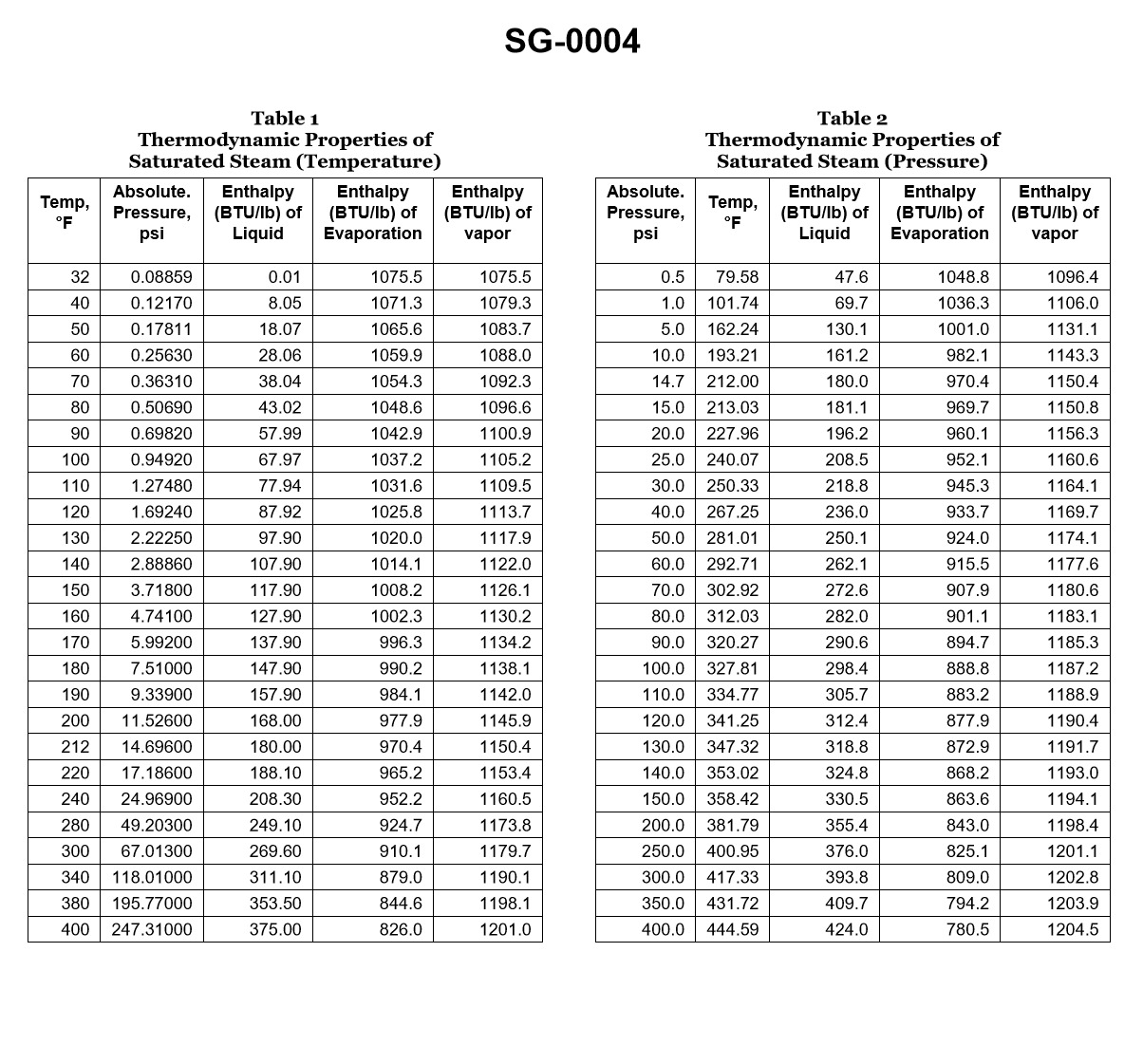
70% to passIf a boiler generates saturated steam at 125.3 psig, how much heat is required to change the water into steam if the feed water temperature is 240°F? Illustration SG-0004
A30.5 Btu/lb
B116.5 Btu/lb
C582.7 Btu/lb
D983.4 Btu/lb
AI Explanation
The correct answer is D) 983.4 Btu/lb. The amount of heat required to change water into saturated steam at a given pressure is known as the latent heat of vaporization. At a pressure of 125.3 psig, the latent heat of vaporization for water is 983.4 Btu/lb. This value can be found in steam tables or other reference materials commonly used for marine engineering calculations. The other options are incorrect because they do not represent the correct latent heat of vaporization for the given pressure. Option A (30.5 Btu/lb) is too low, Option B (116.5 Btu/lb) is also too low, and Option C (582.7 Btu/lb) is significantly lower than the actual value.
Related Questions
Q116:In accordance with Coast Guard Regulations Q243:Serious tube leaks in the air ejector condenser assembly will cause ____________... Q12:The primary function of a flame safeguard system, as used on an automatically fi... Q348:Prior to rolling the main turbines in preparation for getting underway, you shou... Q50:Which of the following locations could desuperheated steam be considered to occu...
Ready to test your knowledge?
Take a Steam Plants - 1st Asst/Chief Practice ExamOfficial Resources